
In this application note, a rapid method for the detection and confirmation of polyphenolic compounds in fruit juices using the ACQUITY UPLC H-Class System is described. With the selectivity of the MS from the ACQUITY SQ Detector, analysis times can be further reduced for quantification of key components.
Polyphenols are widely recognized as functional components of food and beverage products, such as tea, wine, and fruits. Polyphenols are reported to play a potential role in protecting human health from degenerative and cardiovascular diseases.1,2,3
Over the years, the determination of natural phenolic compounds has proved an extremely difficult analytical challenge, which has been hindered by the complexity of the numerous chemical classes and species, combined with inherent limitations of conventional HPLC separation methods.4 Typical HPLC methods that are used to separate and identify polyphenolic compounds by reversed phase chromatography often take more than an hour for a single run.5
More recently, analysis methods of polyphenols using Waters ACQUITY UPLC System have been developed, which provide increased resolution and shortened analysis times.3,6,7 The reduction in run time enables an increase in sample throughput, which is crucial for juice QC laboratories to deliver their products to store shelves more quickly.
Waters ACQUITY UPLC H-Class System enables routine HPLC users to move to UPLC Technology easily by combining the speed and performance of UPLC with the ability to run HPLC separations. The use of the ACQUITY H-Class with UV and mass spectrometry provides a robust solution for the QC analysis of juice samples.
In this application note, a rapid method for the detection and confirmation of polyphenolic compounds in fruit juices using the ACQUITY UPLC H-Class System is described. With the selectivity of the MS from the ACQUITY SQ Detector, analysis times can be further reduced for quantification of key components.
Seven commercially available fruit juice samples, together with 11 polyphenol standards were analyzed.
Each juice sample was filtered through a 0.45 μm filter and diluted with an equal amount of water.
|
LC system: |
ACQUITY UPLC H-Class |
|
Mobile phase A: |
Water + 0.1% acetic acid |
|
Mobile phase B: |
Acetonitrile + 0.1% acetic acid |
|
MS System: |
ACQUITY SQ Detector |
|
Ionization: |
Electrospray |
|
Capillary voltage: |
3.0 kV |
|
Sampling cone: |
30 V |
|
Extraction cone: |
3.0 V |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
500 °C |
|
Desolvation gas: |
1000 L/hr |
|
Cone gas: |
20 L/hr |
|
Scan mass range: |
50 to 650 m/z |
|
UV range: |
210 to 400 nm (Extracted at 280 and 305 nm) |
|
Sampling rate: |
20 pts/sec |
|
Filter time constant: |
Fast |
All data were acquired and processed using Empower 2 Software.
Two methods were developed for the analysis of polyphenols on the ACQUITY UPLC H-Class System. Method 1 is ideal for chromatographic fingerprinting, offering increased separation resolution resulting from a longer column. For QC laboratories performing routine quantification of the key phenolic compounds present, Method 2 would be the preferred method, utilizing a shorter run time, thereby improving lab productivity.
|
Column: |
ACQUITY UPLC HSS T3 2.1 x 100 mm, 1.8 μm |
|
Column temp: |
45 °C |
|
Flow rate: |
0.65 mL/min |
|
Total run time: |
15.0 min |

|
Column: |
ACQUITY UPLC HSS T3 2.1 x 50 mm, 1.8 μm |
|
Column temp.: |
45 °C |
|
Flow rate: |
0.80 mL/min |
|
Total run time: |
7.0 min |

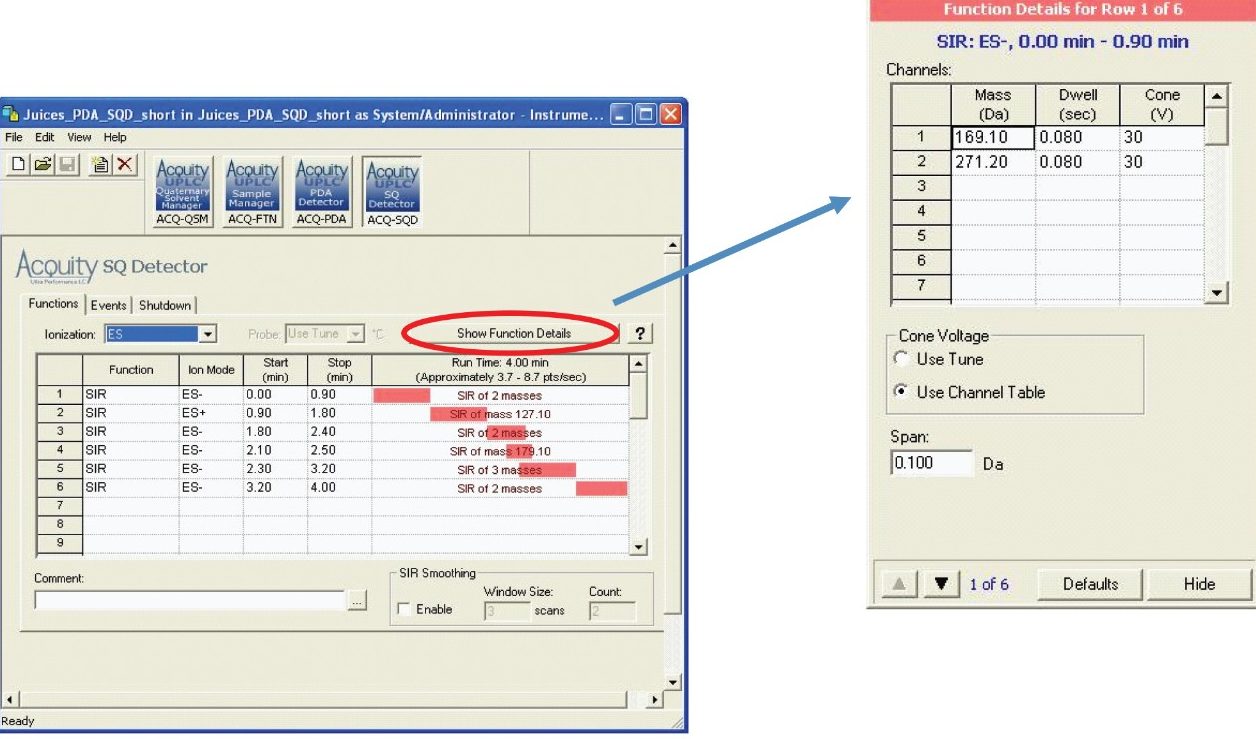
IntelliStart Software was used to automatically develop SIR acquisition methods for the 11 phenolic compounds targeted in this analysis. IntelliStart requires only the entry of basic compound information; then it automatically locates the precursor ion and optimizes the cone voltage, as shown in Table 1. The dwell times were optimized to give a minimum of 15 points across each chromatographic peak for reproducible quantitation. Quantitation of the juice samples was performed by calculating against a calibration curve for each of the polyphenol analytes in solution.

Figure 1 shows a typical MS method setup in Empower Software.
Using the ACQUITY UPLC H-Class System coupled with ACQUITY UPLC PDA and ACQUITY SQ detectors, 11 polyphenol standards were separated and analyzed in 10 minutes, as shown in Figure 2. The PDA result was extracted at wavelengths 280 nm, shown in Figure 2, and 305 nm (data not shown), and SIR chromatograms for the 11 analytes were overlaid, as shown in Figure 2.

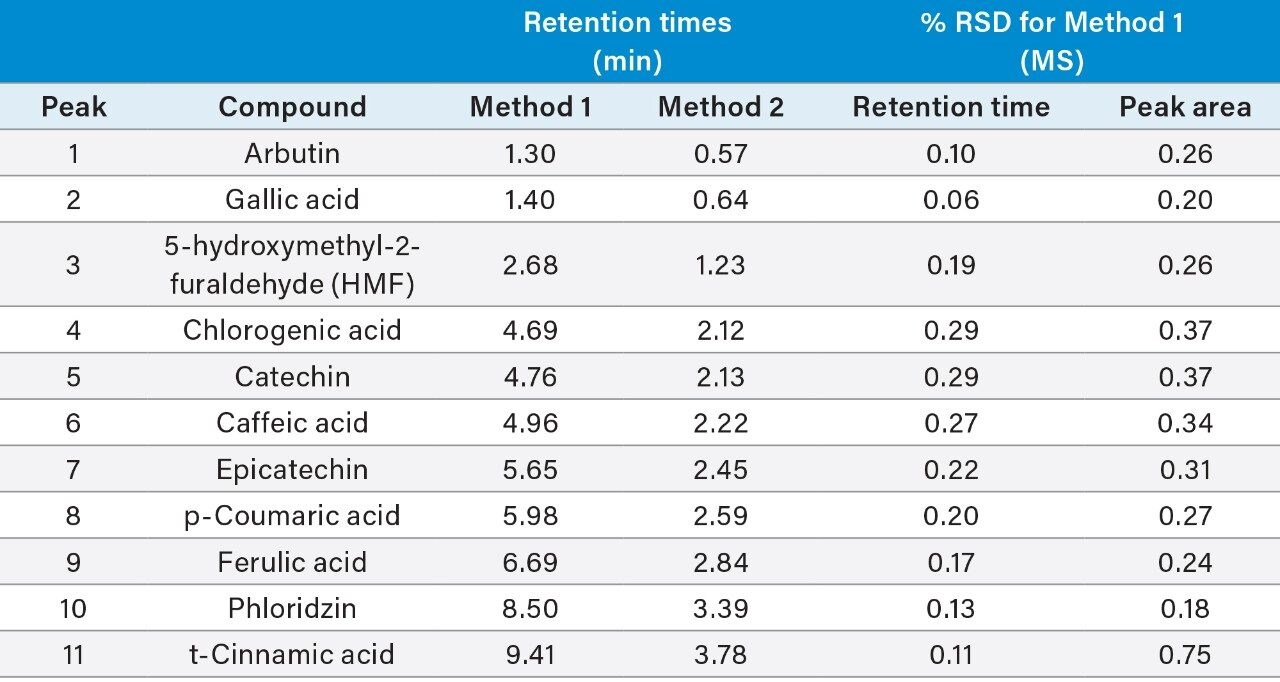
The retention times (Method 1 and Method 2) of the polyphenolic compound standards, along with their retention times and peak area %RSDs (based on six replicate injections) for MS detection using Method 1 are shown in Table 2.
A variety of commercially available fruit juices, namely apple, orange, berry, guava, mango, and lime were analyzed for their polyphenolic content. Both UV and MS data were simultaneously acquired.
An example of the results from an apple juice sample analyzed using Method 1 is shown in Figure 3.

By examining the PDA chromatogram in Figure 3, the polyphenol compounds present in apple juice can be easily assigned and identified by Empower Software.
To increase confidence in component identification beyond simple retention time matching, UV spectral library functions with PDA and Empower Software can be employed. Using HMF (Peak 3) to illustrate, Figure 4 shows the spectrum index plot for HMF standard, and the peak detected at the same retention time in apple juice. The spectral match between the HMF standard and Peak 3 in apple juice confirms the identity of HMF in the apple juice.
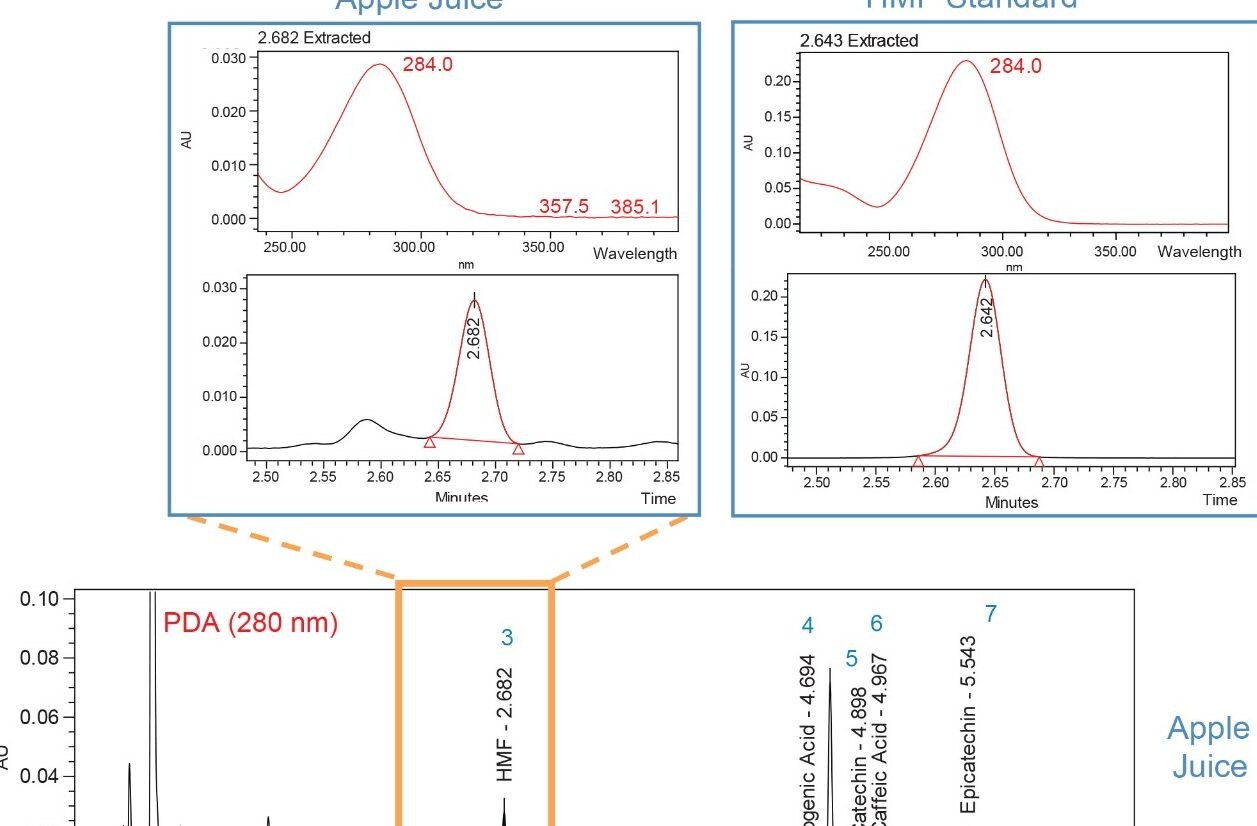
With the additional coupling of a more selective detector, such as the ACQUITY SQ Detector, more information can be obtained in a single sample injection. This provides QC analysts with increased confidence in data interpretation and compound confirmation. From Figure 5, the HMF peak (Peak 3) detected with MS further confirms the polyphenol identity in the juice.
A closer look at the apple juice results in Figure 5 revealed differences in the UV and MS chromatograms.

Catechin (Peak 5), which was assigned and identified using UV retention times, was not detected and quantified by MS (as indicated by the red trace). Identification by UV alone might lead to an incorrect assignment of the polyphenol present, thereby causing mistaken identity of the fruit. The ACQUITY SQ Detector, with its greater specificity, can provide more confidence in data interpretation over a PDA detector alone.
The ACQUITY SQ Detector can be easily added to routine QC laboratories that already use the ACQUITY UPLC H-Class System with Empower Software. Empower users can thereby reap the benefits of MS without the need for additional training. IntelliStart (an MS software component in Empower) enables easy setup and system monitoring of the SQ Detector. It also can automate the method development of selected ion recording (SIR) channels for new compounds.
Another advantage of the MS system is the ability to monitor SIR channels for each of the compounds in the target analysis list. Using the ACQUITY SQ Detector in this mode of operation allows the detector to be selective, and for the user, it is very easy to see whether a specific peak is present or absent in each sample analyzed. Furthermore, an additional benefit that results from the ACQUITY SQ Detector’s selectivity is that the runtime can be reduced, since it is less important to obtain chromatographic resolution (compared to the UV detector) for each of the targeted and matrix peaks. For quantification of key components only, the analysis time was shortened from 10 min to 4 min, (Method 2), which improved sample throughput, as shown in Figure 6.

The polyphenol compounds identified and quantified by MS in the juice samples analyzed are listed in Table 3.

In this work, the ACQUITY UPLC H-Class System, coupled with ACQUITY UPLC PDA and ACQUITY SQ detectors, enabled the separation, detection, identification, and quantification of polyphenols in fruit juices.
Overall, the use of both PDA and MS detectors brings complementary benefits for QC laboratories doing routine QC analyses.
720004069, August 2011