In this application, a fast analytical method is established based on the two-dimensional liquid chromatography to simultaneously assay the contents of vitamin A, α/β/γ/δ-tocopherol, vitamin D2, and vitamin D3 in infant milk powder. The method is simple, fast, highly automated, precise and accurate. It is suitable for the assay of vitamin A, α/β/γ/δ-tocopherol, vitamin D2, and vitamin D3 in formula milk powder or other milk products.
Vitamins A, D, and E are fat-soluble vitamins essential for the body to maintain normal metabolism and functions. Vitamin A, also known as retinol, plays an important role in promoting body growth, maintaining the integrity of the epidermis, etc. Vitamin D includes two major forms, i.e. vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol), which promote calcium and phosphorus metabolism and bone formation in mammals. Vitamin E includes tocopherols and tocotrienols. There are 8 active forms of vitamin E due to the variation of methyl substitution on the parent tocopherol and tocotrienol ring, including α-, β-, γ-, and δ-tocopherols. Among them, α-tocopherol is usually singled out in food science as it has the highest activity and antioxidative and anti-aging properties.1 Infant formula and adult nutritional products and animal feeds are two important forms of fat-soluble vitamin fortified products. In actual samples, vitamin A and vitamin E can be quantified directly because their content levels are relative high and matrix interference is negligible; however, vitamin D is generally added in a small amount, has relatively low UV absorption, and suffers severe interference from the matrix, so liquid chromatography-mass spectrometry or semi-preparative normal-phase clean-up is specified for separation and assay of vitamin D in the current national standards.2 However, the liquid chromatography-mass spectrometry is relatively expensive; while, the normal-phase purification is susceptible to mobile phase conditions and its process is relatively tedious and time-consuming.
This application is based on Waters' existing solutions of two-dimensional ultra-high performance liquid chromatography (UPLC 2D).3 Using ACQUITY Arc (UHPLC) 2D technology under reversed-phase conditions, the separation and assay of vitamin A, α, β, γ, δ-tocopherol, vitamin D2, and vitamin D3 can be completed simultaneously with one sample injection, and the entire assay only takes 15 min. Vitamin A and α, β, γ, δ-tocopherol are separated and quantified on the 1st dimension column, while vitamin D is heart-cut into the trap column after preliminary separation and clean-up on the 1st dimension column, followed by transfer into the 2nd dimension column for further separation and assay to achieve baseline separation of vitamin D2 and D3.
Through the matrix spiking tests done on actual samples provided by relevant companies and the analysis of QC samples, the results showed good linearity, excellent correlation coefficient and excellent repeatability. For five replicated injections, the RSD of retention time was <0.5%, the RSD of peak area was <2%. The limit of detection of vitamin D (D2 and D3) was as low as 0.5 ug/kg.
|
Instrument system: |
Waters ACQUITY Arc 2D system: QSM1 (quaternary pump 1) + QSM2 (quaternary pump 2) + FTN injector + Column Manager (CM-A) equipped with 2-position 6-port valve + 2998 PDA Detector |
||
|
Mobile phase: |
A: water; B: acetonitrile; C: methanol |
||
|
1st dimension analytical column: |
Poroshell 120 PFP (4.6 × 100 mm, 2.7 μm) |
||
|
2nd dimension analytical column: |
XSelect HSS C18SB (3.0 × 150 mm, 3.5 μm) |
||
|
Trap column: |
XBridge BEH C18 Direct Connect HP Trap Column (2.1 mm × 30 mm, 10 μm) |
||
|
Column temp.: |
1st dimension column 35 °C, 2nd dimension column 40 °C |
||
|
Detection wavelength: |
1st dimension VA: 325 nm (0–5.5 min); VD:264 nm (5.5–6.8 min); VE:294 nm (6.8–10 min); 2nd dimension VD2 and VD3: 264 nm (10–15 min) |
|
|
|
Injection volume: |
10 μL |
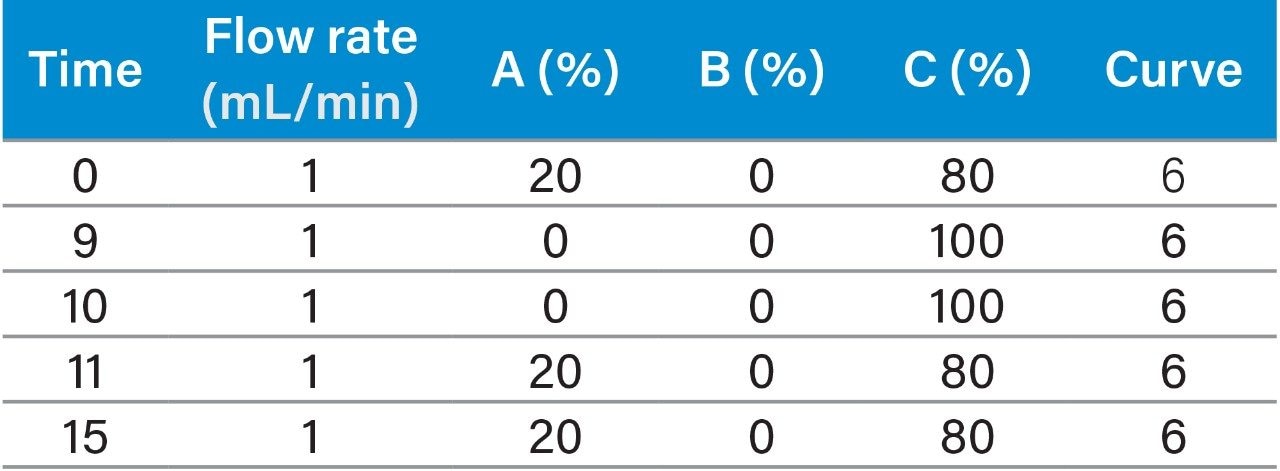
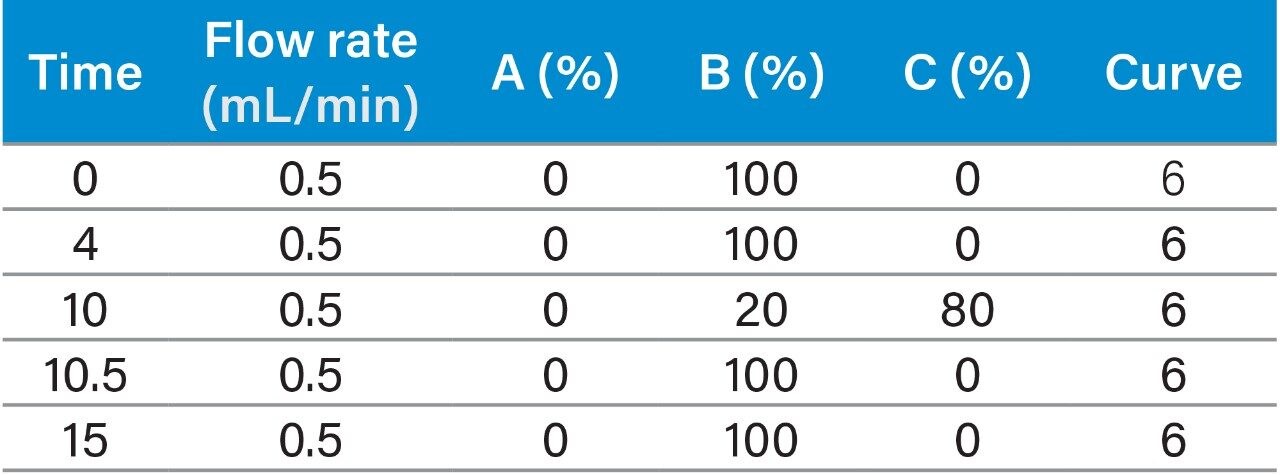
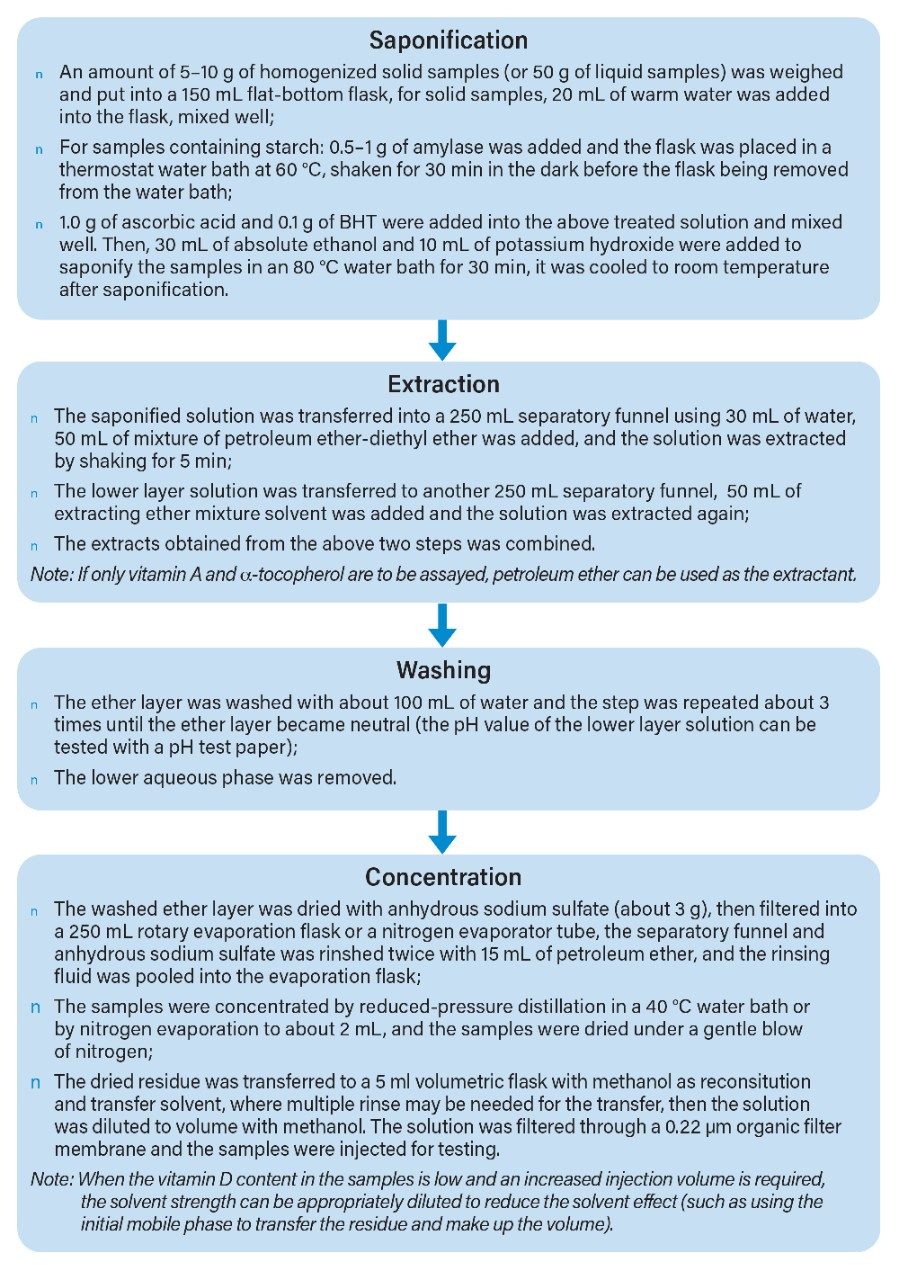
In this application, the test samples were actual samples of milk powder, and sample supply and preparation were provided by our collaborating laboratory. The sample preparation steps were performed using the “GB 5009.82-2016 Assay of Vitamins A, D, and E in Food”2 as a guide. The specific process is as follows:
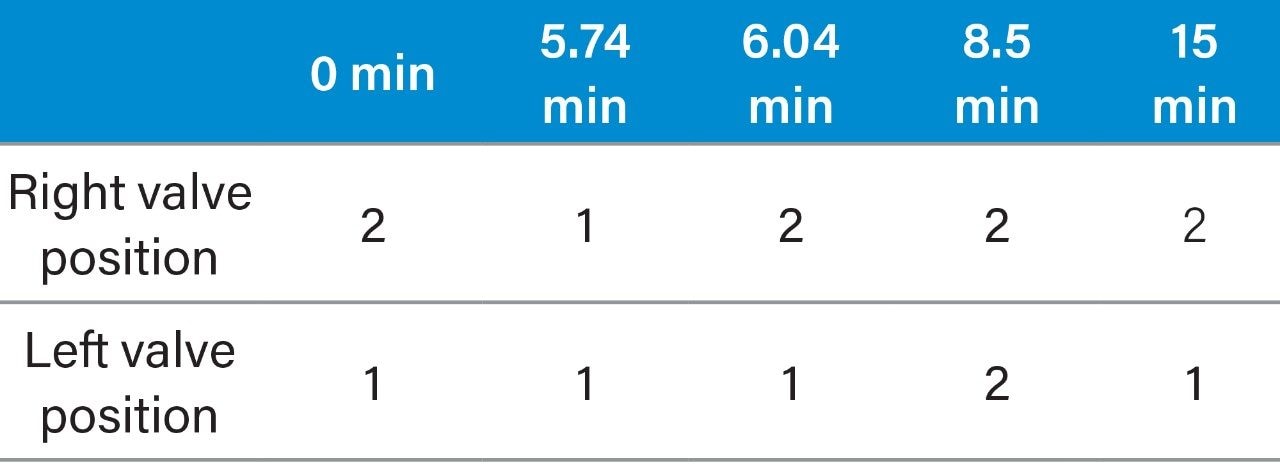
Vitamin A and vitamin E were measured on the 1st dimension LC, and vitamin D was measured on the 2nd dimension LC after heart-cutting. The key to the method is that the 1st dimension LC should be able to separate the vitamin A and vitamin E from the matrix inferference peaks, and it also should have excellent retention time repeatability to ensure the accuracy of heart-cutting with minimal cutting window. In order to more intuitively determine whether the cutting is accurate, and to allow a timely and convenient adjustment in the retention time when it is needed, two 2-position 6-port valves were used to connect a detector, so that both the 1st dimension and the 2nd dimension peaks were collected on the same chromatogram. The method was as follows: Vitamins A, D and E were passed through the 1st dimension column and the detector, and all peaks showed up. At the same time, vitamin D was subjected to heartcutting so that it was separated again on the 2nd dimension column and the peaks were detected again.
The specific configuration used in this application is shown in Figure 2.
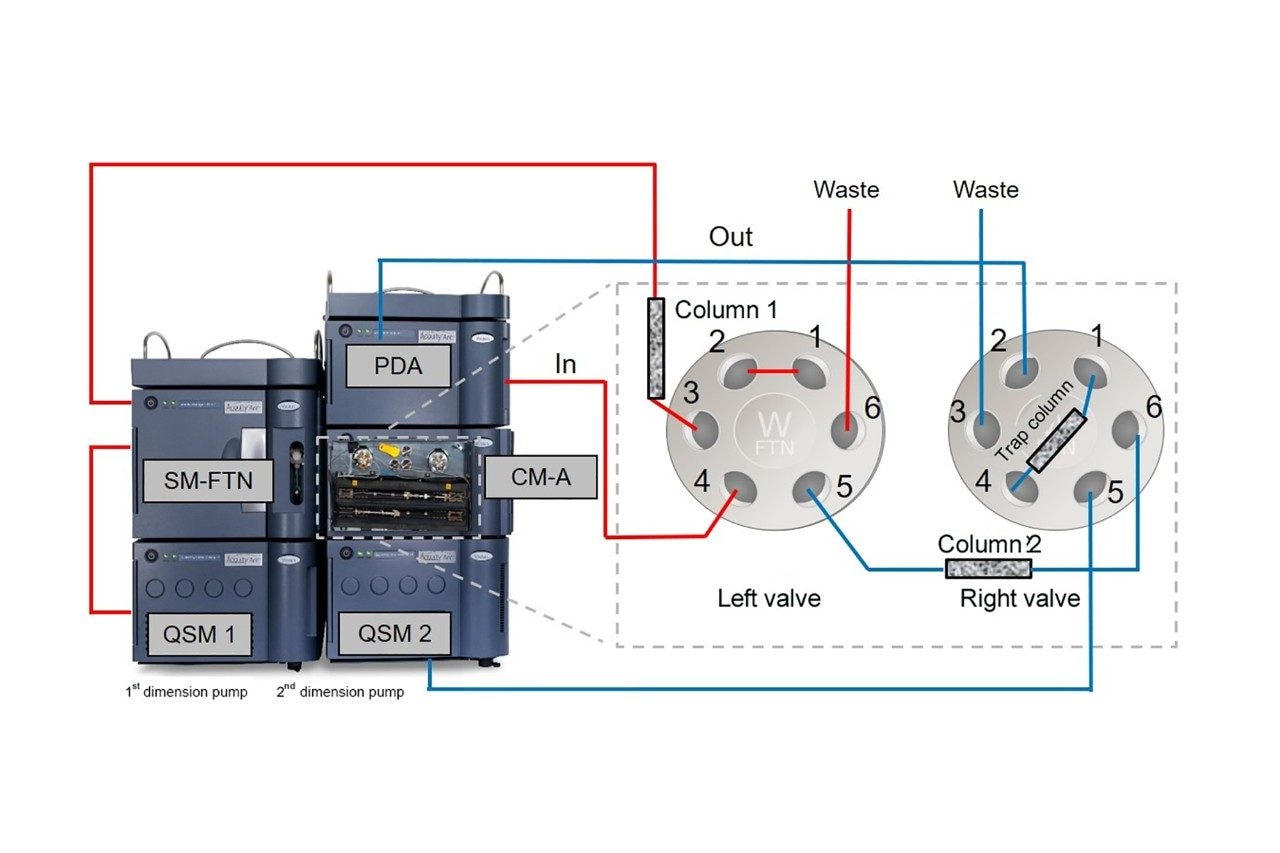
The goal of the 1st dimension separation is to ensure that the vitamin A and the four isoforms of vitamin E are separated and quantified without any interference from matrix. In addition, the vitamin D peak needs to be completely separated from the vitamin A and vitamin E with peak width as narrow as possible, so that it can be completely heart-cut and trapped without any extra amount of matrix background being also cut and transferred to the 2nd dimension, which could affect the further separation of vitamin D on the 2nd dimension LC. The advantage of this method’s connection and configuration is that by using only one detector, one can flexibly adjust the heart-cutting time window based on the retention time of vitamin D in different samples on the 1st dimension LC, so as to avoid any loss of the target substance during the heart-cut process. The goal of the 2nd dimension LC is to not only separate the vitamin D peak from matrix interferences, but also separate the vitamin D2 and the vitamin D3 at baseline resolution in order to achieve qualification and accurate quantitation purposes. Also, the vitamin D2 and D3 peaks should be as far away as possible from the pressure disturbing peaks that is caused by valve switching, as well as other major interfering peaks to ensure the accuracy of quantitation.
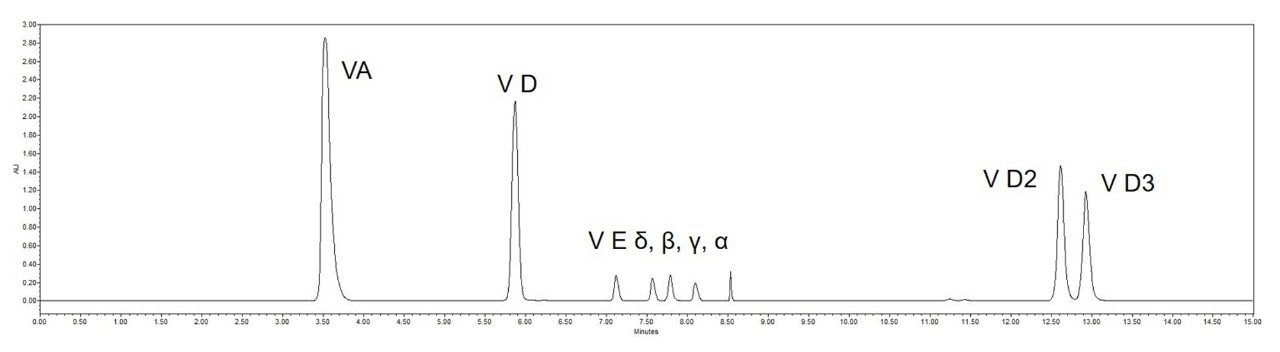
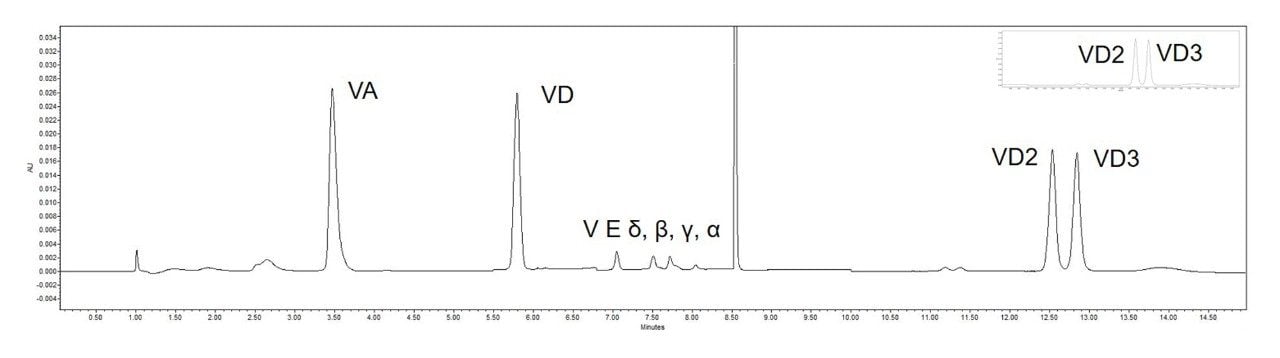
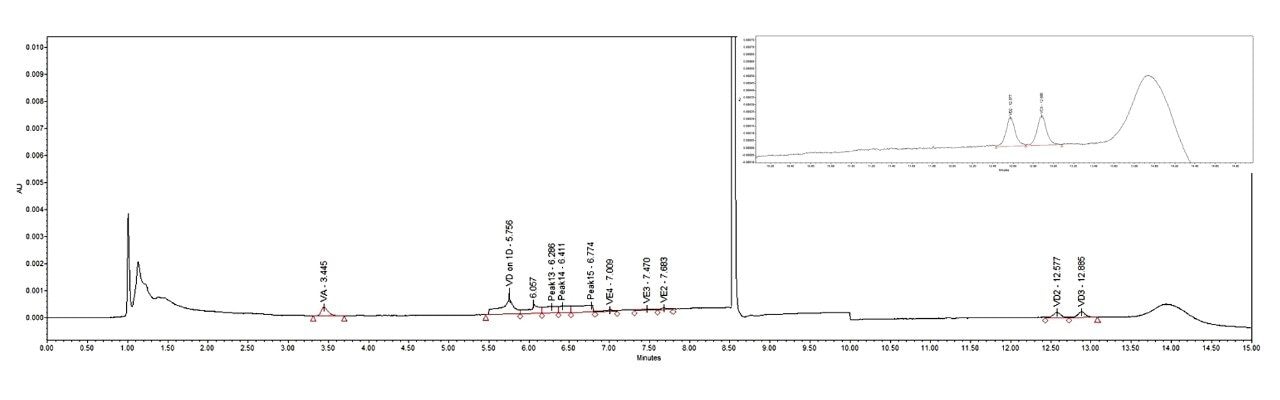
From the separation chromatogram of the standards in Figure 3 and the chromatogram of the actual spiked sample in Figure 4, it can be seen that good separation of vitamin A, four vitamin E isoforms and impurities can be achieved on the 1st dimension column, and good separation of two vitamin Ds and impurities can be achieved on the 2nd dimension column.
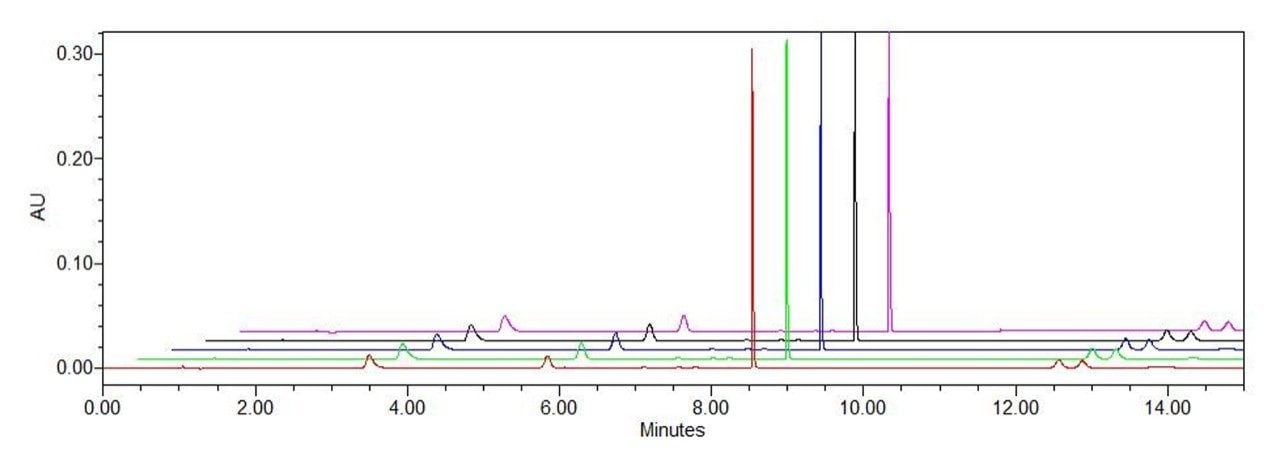
The results of repeated injections (n = 5) of the standards at a 5 μg/L concentration are shown in Figure 5. The RSD for the retention time of vitamin A and four vitamin E isoforms on the 1st dimension LC and the RSD for the retention time of vitamin D2 and vitamin D3 on the 2nd dimension LC was all <0.5%, and the RSD for their peak area was <2%, which ensures the accuracy of the heart-cutting method.
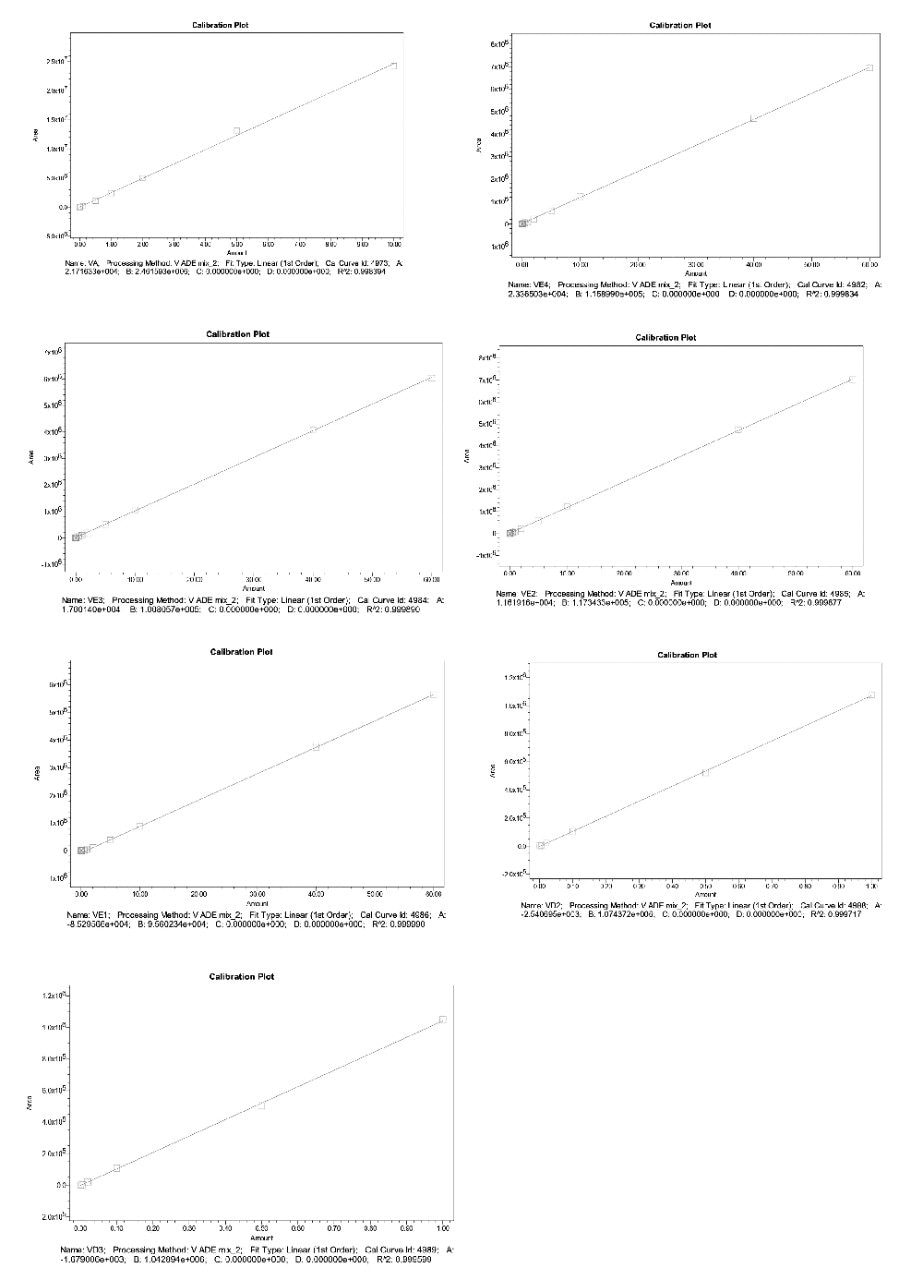
In this application, the linearity of vitamin A, four vitamin E isoforms, vitamin D2, and vitamin D3 was examined on the basis of the requirements specified in the national standards. The results are shown in Figure 6. Different vitamins all maintained good linear relationships in different linear ranges, and the coefficient of determination R2 was greater than 0.998.
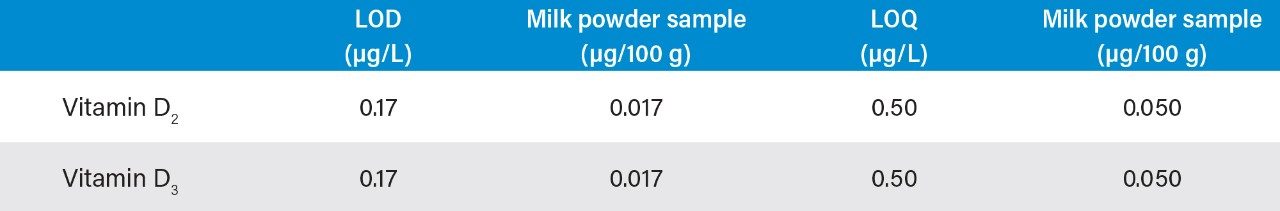
In actual samples, the contents of vitamin A and vitamin E are generally high and can be accurately quantified, while the content of vitamin D is generally low and is prone to matrix interference, and hence the quantitation of vitamin D is relatively difficult. Therefore, the LOD of vitamin D is an important parameter in the evaluation of this instrument method. Detection sensitivity was investigated in this work. When the injection volume was 250 μL, the measured S/N ratio of vitamin D2 and vitamin D3 was 11 at an injection concentration of 0.5 μg/L, as shown in Figure 7. The refore, 0.5 μg/L was used as the LOQ of vitamin D. The LOD of vitamin D was calculated at 0.17 μg/L using a S/N of 3. This LOQ meets the assay requirement for the lowest concentration point of 50.0 μg/L on the standard curve in liquid chromatography methods specified in national standards. The LOD and LOQ results are shown in Table 1. Therefore, if the content of vitamin D in the sample is relatively high, an injection volume of 10 μL can be used to meet the requirements described in national standards.
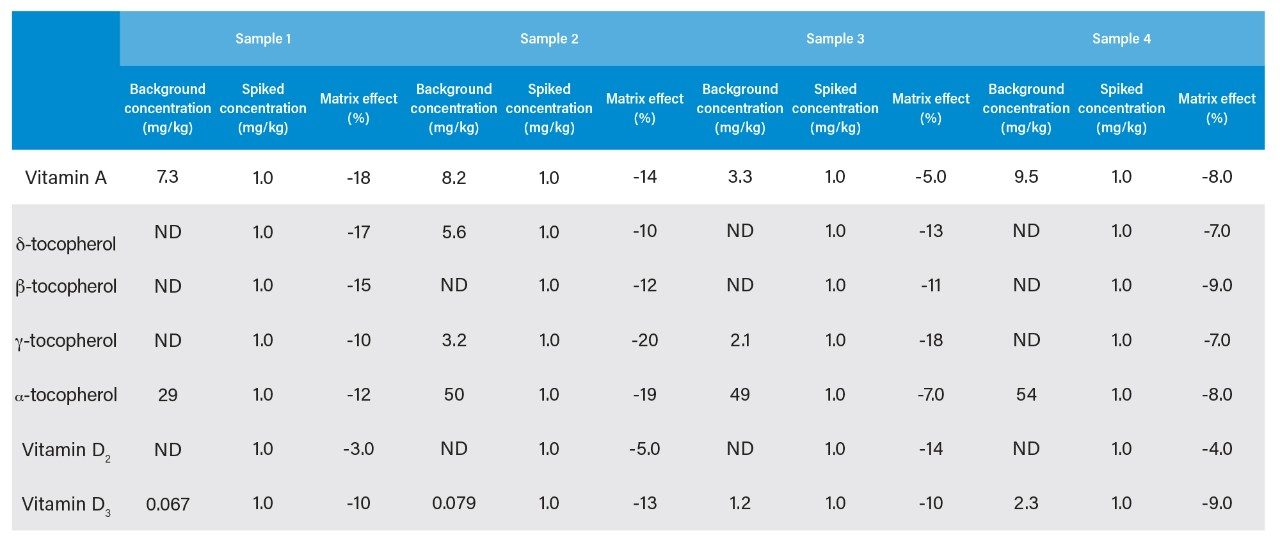
Using the method established in this application, milk powder samples provided by our clients were analyzed. Sample preparation was performed by the client according to the preparation method described in this document. The results show that the vitamins added to this batch of samples are mostly vitamin D3 and α-tocopherol (Table 2).
The magnitude of the matrix effect has a direct impact on the quantitative results. This application also evaluated the matrix effect at the same time. The above samples provided by the client were also spiked and the test results were used as the basis for evaluating the matrix effect. The test results of the actual samples and the evaluation of the matrix effect are shown in Table 2.
In this application, a fast analytical method is established based on the two-dimensional liquid chromatography to simultaneously assay the contents of vitamin A, α/β/γ/δ-tocopherol, vitamin D2, and vitamin D3 in infant milk powder. The method is simple, fast, highly automated, precise and accurate. It is suitable for the assay of vitamin A, α/β/γ/δ-tocopherol, vitamin D2, and vitamin D3 in formula milk powder or other milk products.
720006655, March 2020